Antivenom Krise
Der Einsatz von Gegengiften ist eine der wichtigsten Maßnahmen zur Kontrolle und Bewältigung von Schlangenbissvergiftungen. Trotz der hohen Prävalenz von Schlangenbissen und des damit verbundenen Bedarfs an wirksamen Gegengiften ist die Verfügbarkeit von Gegengiften zurückgegangen. Die letzten Jahrzehnte waren durch einen Rückgang der Produktion, einen Vertrauensverlust in die Produkte und die unzureichende Nutzung bewährter lebensrettender Gegengiftprodukte gekennzeichnet.
Die Problematik des Schlangengegengifts
Da ärmere und marginalisierte Bevölkerungsgruppen besonders von Schlangenbissen betroffen sind, ist der Marktanreiz für die Erforschung und Herstellung von Gegengiften sehr gering. Außerdem sind Gegengifte sehr spezifisch für eine oder wenige Schlangenarten und werden daher nur für eine bestimmte Populationsgröße verwendet, was die Forschung und Produktion noch unattraktiver macht.
Dies liegt daran, dass die Toxine, die bei einem Schlangenbiss in den Körper gelangen, von Schlangenart zu Schlangenart sehr unterschiedlich sind und auch regionale Unterschiede aufweisen können. Daher müssen Gegengifte für jede Schlangenart einzeln hergestellt werden, das sogenannte monovalente Antivenom, oder für mehrere Schlangenarten in einem Gegengift gemischt werden, das sogenannte polyvalente Antivenom. Dazu müssen die regional relevanten Schlangenarten und deren Auswirkungen bekannt sein. Nicht nur die Herstellung ist kompliziert, sondern auch die klinische Prüfung und Registrierung der Gegengifte in den einzelnen Ländern ist teuer und zeitaufwändig. Viele Antivenome, die auf dem Markt sind und verwendet werden, haben keine präklinischen oder klinischen Ergebnisse.
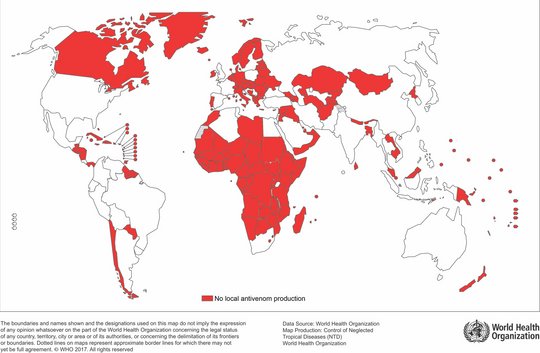
Daher wäre eine einheitlichere Regelung und Durchführung von klinischen Studien und Zulassungen von Vorteil. Für den afrikanischen Kontinent könnte die neu eingeführte Afrikanische Arzneimittelagentur (AMA) mit Sitz in Ruanda ein Weg sein, dies zu verbessern. Außerdem wird nur ein kleiner Teil der Gegengifte für den internationalen Gebrauch hergestellt, während die meisten Hersteller nur für den nationalen oder regionalen Gebrauch produzieren. Dies zeigt, wie wichtig die lokale Produktion ist, die insbesondere in Ländern, in denen Schlangenbisse weit verbreitet sind, gefördert werden sollte. Wie aus der Karte auf der rechten Seite hervorgeht, ist dies vor allem auf dem afrikanischen Kontinent notwendig, wo Schlangenbisse weit verbreitet sind und die lokale Produktion gering ist.
Der "Teufelskreis"
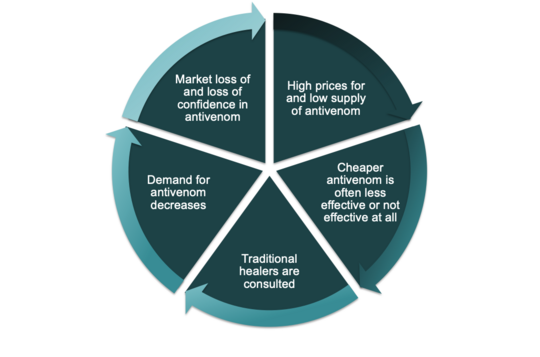
Warum diese Krise andauert und wie sie zu dem bereits erwähnten Vertrauensverlust und der unzureichenden Nutzung von Antivenom-Produkten führt, lässt sich durch den "Teufelskreis" erklären. Die Ausgangssituation ist ein geringes Angebot an Gegengift und infolgedessen hohe Preise für Gegengift. Dies führt häufig dazu, dass billigere - und oft weniger wirksame oder sogar gefälschte - Gegengifte zur Behandlung von Patienten und Patientinnen mit Schlangenbissvergiftungen eingesetzt werden. Dies führt zu einem Vertrauensverlust in Gegengiftprodukte im Allgemeinen und zu einer unzureichenden Verwendung von Gegengift. Bei einem Schlangenbiss suchen die Patienten und Patientinnen eher traditionelle Heiler:innen auf, und die Nachfrage nach Antivenom geht weiter zurück. Dies wiederum führt zu einem weiteren Marktverlust für die Anbieter von Antivenom, was die Preise noch weiter in die Höhe treibt, während das Angebot weiter zurückgeht.
Lösungsansätze
Es gibt eindeutig keine Einzellösung für die Antivenom-Krise, sondern eher eine Mischung von Maßnahmen, die sich gegenseitig unterstützen. Zwei der oben genannten Maßnahmen sind eine verstärkte lokale Produktion von Antivenom und eine verstärkte koordinierte Zulassung von Antivenom. Das auf dem Markt befindliche Gegengift muss seine Wirksamkeit in präklinischen und klinischen Versuchen nachgewiesen haben. Dies erfordert auch eine Stärkung der Forschungskapazitäten im Allgemeinen.
Dies muss natürlich Hand in Hand mit der Ausweitung der universellen Gesundheitsversorgung (Universal Health Coverage, UHC) gehen, die im Idealfall die Kosten für die Behandlung abdecken würde. Logistische Innovationen können ein Hebel sein, um UHC zu erreichen und Menschen überall zu erreichen und zu behandeln.